Latest AnaBios Webinar
Human Ventricular Action Potential and E14 Waiver for Novel Drugs
Presented by Dr. Derek Leishman (Eli Lilly & Company) & Dr. Najah Abi-Gerges (AnaBios)
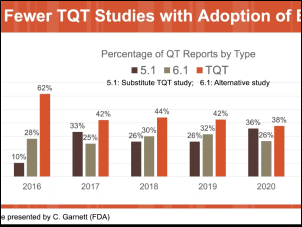
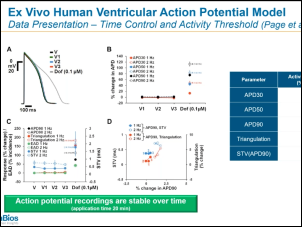
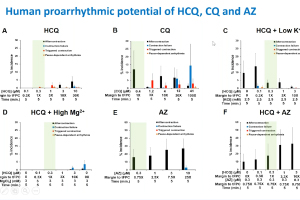
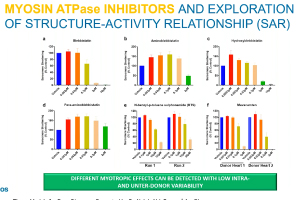
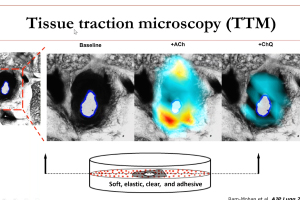
In 2013, the FDA first presented the Comprehensive In Vitro Proarrhythmia Assay (CiPA) initiative as an upgrade to S7B/E14 guidance from 2005. In the past 10 years, the CiPA initiative has led to a commonsense approach to hERG testing. Pharma companies testing compounds with hERG block or borderline hERG block have the option of conducting additional ion channel studies, in silico modeling and/or testing with cardiomyoctyes (either stem cell-derived or primary human cardiomyoctes) as described in the most recent FDA Q&A documentation. E14 guidance points toward a thorough QT (TQT) study during Phase II clinical trials that is both time consuming and expensive (i.e., greater than $1M). With CiPA, sponsors may request a waiver of the TQT study in exchange for an assessment of EKG’s during Phase I and adequate preclinical data. During this webinar, Dr. Najah Abi-Gerges (VP, R&D at AnaBios) discusses a ventricular action potential assay provided by AnaBios that involves heart tissue from ethically consented human donors that can contribute to the preclinical data set for E14 waiver requests. Dr. Derek Leishman (VP, Translational & Quantitative Toxicology at Eli Lilly) presents a big pharma perspective on E14 waiver requests and relevant strategies for safety pharmacology in general.